Regulation ready expertise
Get distilled insights and analysis from experts in life sciences and food regulation, so you can see more, faster.
Food Regulatory Update: State Activity in 2026
More FDA leaders are departing. Here’s what the turmoil may mean for agency policies and priorities
Everything you need to understand, plan for, and shape regulation
AgencyIQ delivers deep, timely, and analytical coverage of regulatory developments shaping life sciences and food products. Our experts provide a daily synthesis of complex policy—helping you understand what’s changing, why it matters, and what comes next.
Stay up to date with personalized alerts and workflows, newsletters, and collaboration tools in one easy-to-use workspace built to help you see the big picture, act faster, and plan your next move with confidence.
Stay up to date
Sign up for our free newsletters to receive insights on regulatory developments in your industry.
FDA Today: Life Sciences
Catch up on impacts and implications that the latest regulations, policies, and actions of the Food and Drug Administrations (FDA) have on the life science industry.
FDA Today: Food
Clear, expert-driven insights into the policies shaping food and food-related chemicals. Get key regulatory updates and analysis from FDA, USDA, EPA, and state agencies—delivered straight to your inbox.
EMA Today
Stay on top of current life sciences regulation in the European market with a quick-to-read summary, supported by thoughtful and exclusive analysis.
One connected workspace for regulatory intelligence
Stay in the know
Daily, concise synthesis of research, analysis, and updates to help you gain clarity and get up to date quickly on developments within your industry.
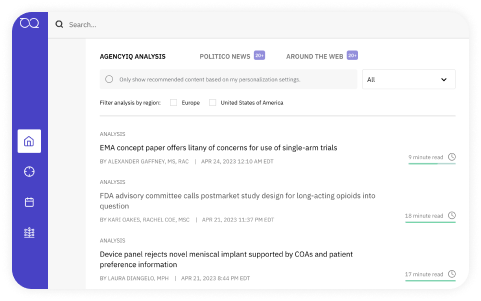

Information that
works for you
Regulatory experts sift through the latest guidance, rules, and legislation—so you don’t have to. Giving you customized insights so you can act fast, predict outcomes, and make an impact. Use the on-platform Ask the Expert feature to interact with our research team directly.
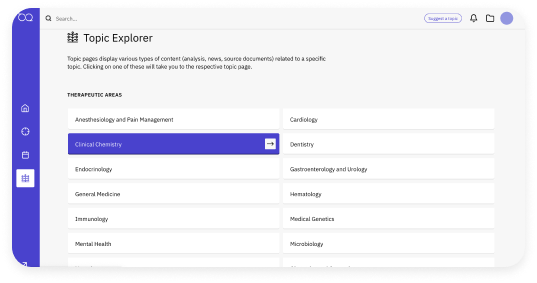

News that impacts you
Daily coverage from a large, non-partisan newsroom helps you gain insights research from the #1 most trusted regulatory intelligence source.
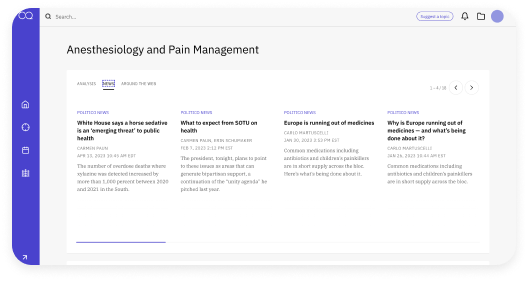

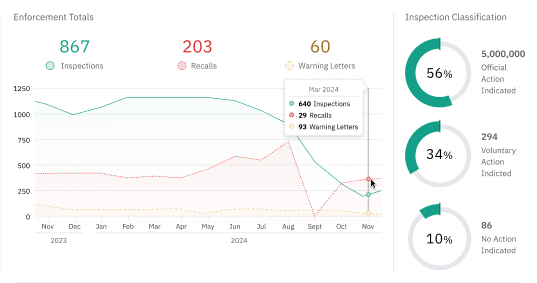
Close the Gap between Policy and Enforcement
Stay ahead of enforcement shifts with complete visibility, right when you need it most. Inspection Navigator gives regulatory teams a faster, smarter way to view FDA inspection trends, access critical 483s, and align compliance strategies—all in one intuitive platform.

Collaborate seamlessly
Create your own collections to organize your regulatory documents and relevant content or take advantage of the Curated Collections that are organized and updated by the AgencyIQ research team on specific policy topics. Collaborate by sharing regulatory research, news, and analysis with your teammates and stakeholders through the Collections feature.

Access, track, and evaluate information
Powerful search tools allow you to quickly retrieve results, notices, warning letters, recalls, and more. Discover regulations that are important to your organization. Access the status of Citizen Petitions submitted to Federal agencies and view a side-by-side comparison to weigh different versions of guidance documents with our easy-to-use Guidance Tracker.

Identify Policy Changes
Monitor bills of selected federal congressional committees and subcommittees to follow legislative developments that could impact your industry (Life Sciences Platform Only).

Regulation insights on
Life Sciences
- Advanced Therapies
- Biologics
- Blood Products
- Diagnostics
- Dietary Supplements
- Generics/Biosimilars
- Medical Devices
- OTC Products
- Pharmaceuticals
Food
- Labeling and Claims
- Additives and GRAS Substances
- Packaging and Contact Materials
- Ingredient and Chemical Safety
- Pesticides and Contaminants
- PFAS and Emerging Risks
- Inspections and Investigations
- Recalls and Outbreaks
- Ultra-processed Food Policy
- Nutrition
Top regulation experts
With more than 100+ combined years of regulatory experience, AgencyIQ regulatory experts deliver accurate and timely insights—ensuring a bird’s-eye view of regulations.



















